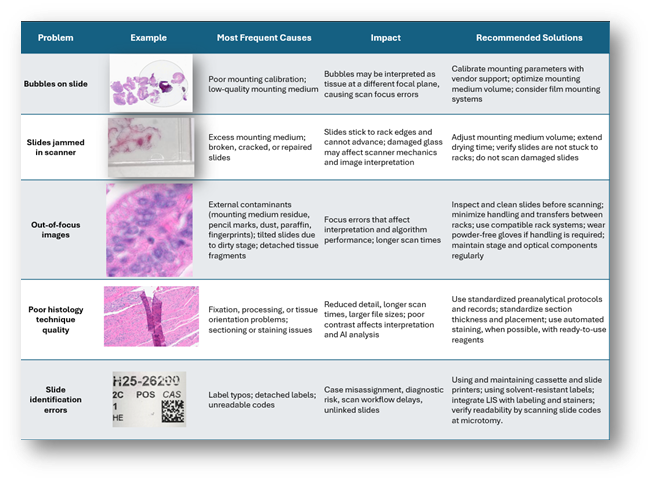
In digital pathology, the preanalytical stage, as part of the histopathological process, encompasses everything that happens to the sample from its collection to digitization. Unlike traditional microscopy, it requires greater rigor, since any technical artifact is amplified during scanning and can affect image quality (Table 1), AI analysis, or workflow efficiency (1).
The accreditation guidelines of the College of American Pathologists (CAP) recommend validating all preanalytical conditions, even from sample collection. Therefore, it is essential to have standardized procedures, formal validations, and robust traceability (2,3). Today, laboratory information systems (LIS) and modern hardware allow for efficient tracking of samples throughout the entire process. The use of electronic request systems, ideally integrated with the Hospital Information System (HIS), facilitates correct identification and appropriate transmission of clinical information (4). Next, we will briefly review the main aspects that comprise the pre-analytical stage and how, at Clínica Alemana in Santiago, Chile, we have adapted them to facilitate the digital transition of the process.
Sample Collection and Transport:
Initial specimen handling determines the quality of all subsequent studies. This step is strongly supported by having a well-structured institutional procedures document that provides clear guidelines for handling each specimen type, describes the processes involved, and defines responsibilities for each task. The document should be sufficiently detailed, practical, and focused on achieving the highest possible level of standardization. Fixation protocols should be clearly regulated within this document, following recognized recommendations such as those from the CAP, including proper execution and event documentation.
Assigning a unique Case ID from the moment of specimen collection and maintaining it throughout the workflow greatly simplifies traceability of all case elements (specimens, blocks, and slides) and prevents relabeling during laboratory accessioning. Labeling should always be performed at the place and time of collection, using barcode or similar systems to enable registration at each processing station. These checkpoints can be integrated into a computerized traceability platform linked to routine manual steps. Grouped transport under a single electronic container identifier is an efficient approach. For intra-hospital logistics, direct pickup by pathology laboratory staff using LIS-generated route lists has proven to be a reliable and well-controlled strategy.
Accessioning and Macroscopic Examination:
Receiving specimens with well-controlled preanalytical conditions gives the laboratory greater confidence in the reliability of downstream results. When upstream control is limited, the reception step becomes critical and must include additional verification checkpoints. At this stage, each container should be reviewed against defined acceptance and rejection criteria by properly trained and experienced personnel. Gross examination data should be recorded in a single digital system to avoid fragmented manual records. Systematic photographic documentation of specimens is highly valuable for retrospective review and diagnostic context, whether integrated into the LIS or implemented locally. Printing uniquely coded histology cassettes individually (rather than in batches) is also a key safety measure to reduce identification errors.
Processing, Embedding, and Sectioning:
Histologic processing is typically performed in batches, so traceability can be managed using coded block baskets, similar to transport workflows. A more robust alternative is one-by-one block tracking at each stage through barcode scanning into the LIS, although this requires more time and an appropriate formalin-safe workspace. Image-recognition devices that read all cassette codes in a basket at once offer a practical, safer compromise, though they require system integration. During embedding and sectioning, workstations should provide access to gross examination data, barcode readers, and dedicated slide printers to support one-to-one processing. Avoiding batch printing and shared printers reduces identification errors. LIS-based tracking of staining baskets can further automate and secure slide progression through each step.
Staining and Mounting:
This automated stage depends mainly on two factors: stable H&E staining and an appropriate slide-mounting system. Using ready-to-use reagents improves staining consistency and saves preparation time. Glass and film mounting systems are both suitable for digital scanning, but each presents specific advantages and limitations that should be weighed during selection and must be properly calibrated with the specific scanning platform in use. Slides prepared externally should be checked carefully, as they may cause focus errors or mechanical issues during digitization.
Table 1. Common Preanalytical Problems in Digital Pathology
Digitization:
At the digitization stage, slides should enter the scanner only after a thorough final quality check. Minimizing handling beforehand is considered best practice. Tray-level controls should verify staining quality, correct slide and rack positioning, smooth slide movement, and overall physical integrity. Integrating the scanner with the LIS allows each slide to be automatically recorded as digitized, closing the traceability chain and completing the preanalytical cycle.
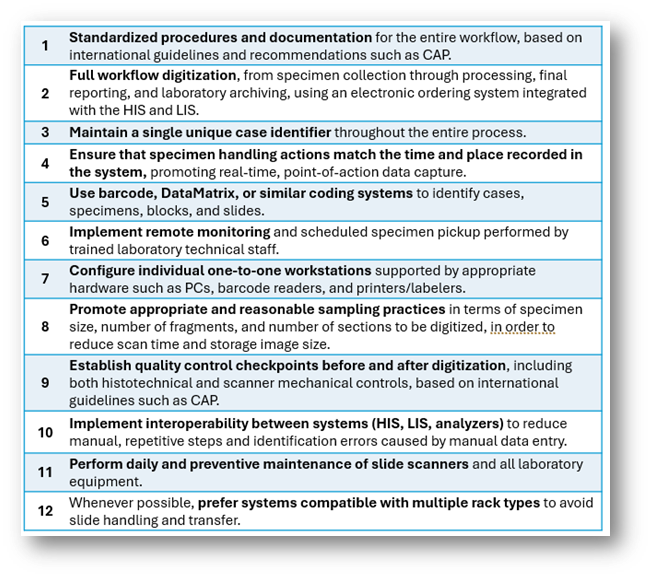
Conclusions and Pre-analytical Best Practices: (Table 2)
Many critical processes must be optimized before scanning begins. Digital scanners require slides with strict technical characteristics to ensure proper focus and artifact-free images. Published evidence shows that introducing dedicated software and hardware for preanalytical management improves efficiency, cost control, and quality in digital pathology programs. In Chile and Latinoamérica, specialized medical technologists or histotechnologists play a central role in executing and supervising these steps, driving process improvements, and implementing informatics tools to secure optimal preanalytical conditions for every patient.
Table 2. Preanalytical Best Practices in Digital Pathology:
References:
- Lujan, G., Quigley, J., Hartman, D., Parwani, A., Roehmholdt, B., Van Meter, B., Ardon, O., Hanna, M., Kelly, D., Sowards, C., Montalto, M., Bui, M., Zarella, M., LaRosa, V., Slootweg, G., Retamero, J., Lloyd, M., Madory, J., & Bowman, D. (2021). Dissecting the Business Case for Adoption and Implementation of Digital Pathology: A White Paper from the Digital Pathology Association. Journal of Pathology Informatics, 12. https://doi.org/10.4103/jpi.jpi_67_20.
- Evans, A., Brown, R., Bui, M., Chlipala, E., Lacchetti, C., Milner, D., Pantanowitz, L., Parwani, A., Reid, K., Riben, M., Reuter, V., Stephens, L., Stewart, R., & Thomas, N. (2021). Validating Whole Slide Imaging Systems for Diagnostic Purposes in Pathology: Guideline Update From the College of American Pathologists in Collaboration With the American Society for Clinical Pathology and the Association for Pathology Informatics. Archives of pathology & laboratory medicine. https://doi.org/10.5858/arpa.2020-0723-cp.
- LaFriniere, M., Bui, M. M., Evans, A. J., Parwani, A. V., Nasim, M., Lott, R., Pantanowitz, L., Routhier, C., Pert, J., Valdez, A., & Trejo Bittar, H. E. (2019). Practical tips to assist implementation of whole slide imaging. College of American Pathologists.
- Fraggetta, F., L’Imperio, V., Ameisen, D., Carvalho, R., Leh, S., Kiehl, T., Serbanescu, M., Racoceanu, D., Della Mea, V., Polónia, A., Zerbe, N., & Eloy, C. (2021). Best Practice Recommendations for the Implementation of a Digital Pathology Workflow in the Anatomic Pathology Laboratory by the European Society of Digital and Integrative Pathology (ESDIP). Diagnostics, 11. https://doi.org/10.3390/diagnostics11112167.
Author

Juan Pablo Pastene de la Fuente
Medical Technologist
Clínica Alemana de Santiago, Chile
PathVisions25 Developing Economy Travel Award Winner
Please log in to your DPA profile to submit comments